
Different scientists contributed their work to the formation of the periodic table.Key Characteristics of the Periodic Table:Įlements are arranged in order of increasing atomic number.Įlements of the Periodic Table are denoted by a unique symbol and not its entire name, as some elements’ names can be long and complex in nature.Įlements are arranged vertically and horizontally. In conclusion, a printable periodic table is a helpful tool in order to learn about elements and solve chemistry problems. Different colors that denote groups of identical elements.The other information that can be included in the cells is The second one is the one or two letter atomic number. It may be present at the top of the cell, in the center, or to the left. The first one is the element’s atomic number. Usually, it contains two pieces of information. F-block: it is placed below the table and contains the lanthanides and actinides.Įach cell includes different pieces of information. 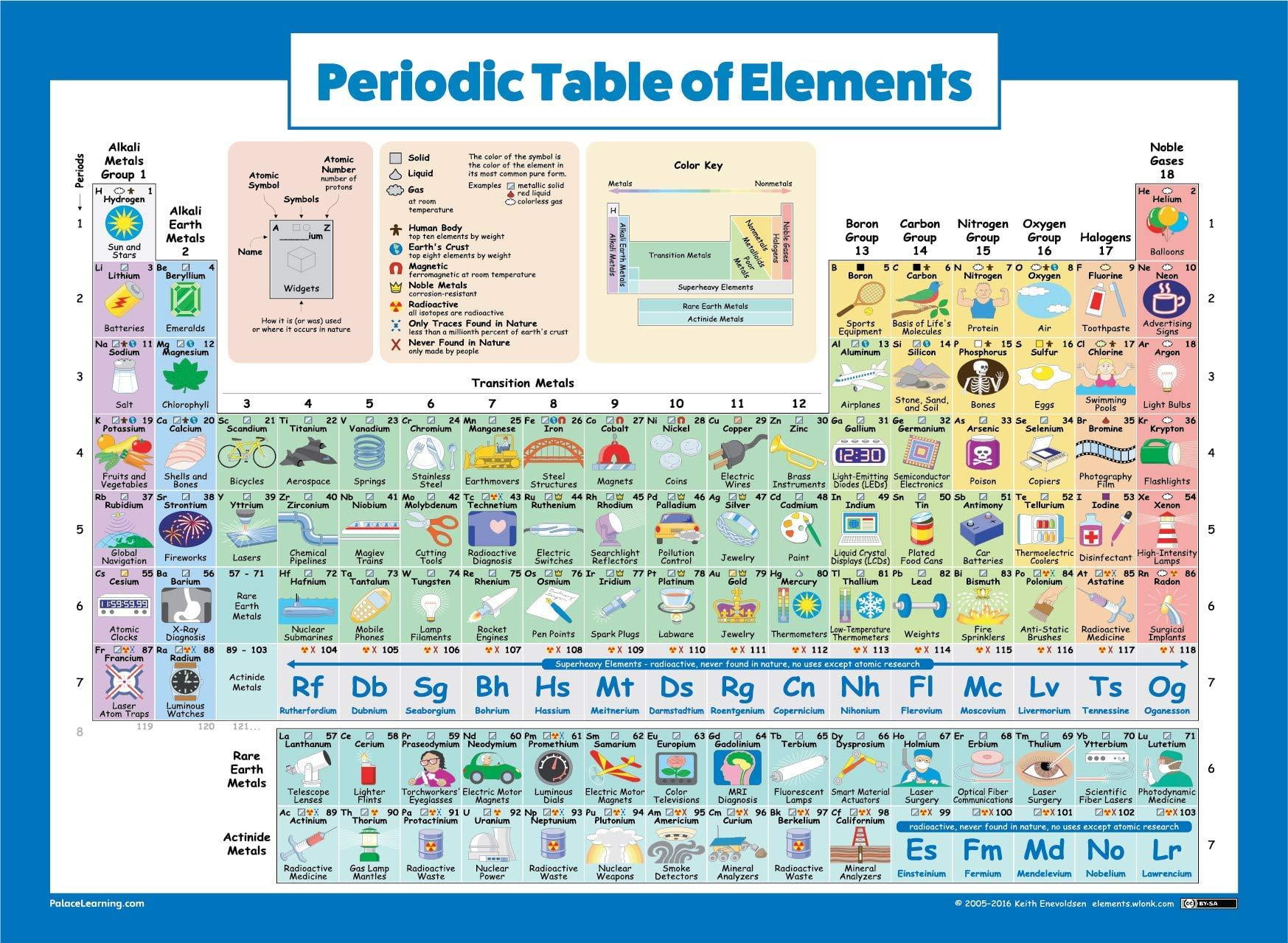
D-block: groups 3-12 are in this block.S-block: It contains alkali metals, alkaline earth metals, helium, and hydrogen.Each block contains different groups, listed below In the periodic table, there are four blocks. The elements that have identical chemical characteristics are assigned a group name and colored the same color. Rows and columns are called periods and groups. Only few people know about these elements because they are unstable and decay rapidly.Ī periodic table consists of cells and each cell represents an element. They were placed at the seventh row off the table. Some of them were officially recognized in 2015. Since the 1940s, scientists discovered he elements that are numbered 104- 118. However, at that time scientist rejected his suggestion but later research proved he was right. In 1945, he published a paper in which he said that actinide series of elements should be placed below the lanthanide series. Seaborg was working on the Manhattan project he hypothesized that additions needed to be made to the periodic table. During the middle of 20th century, when scientists discovered protons then an element assigned atomic number on the basis of the number of protons it contains. He published the 18 column periodic table in 1923. And, second it allowed future scientists that without damaging the integrity of table, they can adjust the position of elements. The first one is that he allowed the placing of new elements and groups of elements he didn’t predicted. Mendeleev also said that elements’ properties can be predicted by their mass and position in the periodic table.Īdditionally, Mendeleev table was important for two reasons due to the addition of dashes.The above feature helped him to realize that some elements were not in the right place by ordering them according to mass.He arranged his table by atomic mass and stated that elements with similar atomic masses had similar chemical properties.He hypothesized the existence of undiscovered elements and used a dash in his table where he assumed those elements should be placed.His table contains the following interesting features Furthermore, he not only documented new elements but also leave space for the discovery of new ones. For the development of the modern periodic table, he is appreciated. In the 1860s, three men Dimitri Ivanovich Mendeleev, Lothar Meyer, and William Odling published periodic tables. However, he was not appreciated at that time but later on and now he is appreciated with the first use of the term periodic in chemistry. The elements that were differed by a factor of 8, he paired them with masses. He was the first scientist who assigns atomic numbers to the elements based on an element’s mass. In the early 1860s, John Newlands divided 62 known elements into groups of eight. Moreover, he classified his elements into two groups i.e. He determined new elements like hydrogen, oxygen, and nitrogen. Antoine-Laurent de Lavoisier:Īntoine-Laurent de Lavoisier wrote the first textbook the Elementary Treatise of Chemistry that was devoted to Chemistry. This definition was used for 3 years until subatomic particles were discovered. He states that it can be something that cannot be broken into smaller parts by a chemical reaction. Some of the people who took part in the formation of the periodic table are Robert Boyle:Īround 1650, the Robert Boyle developed the definition of an element. 8 What is interesting thing to know about the periodic table?.7 How the elements of period table are classified?.5 What are the 7 periodic table groups?.2 How to use a printable periodic table?.1 History of the printable periodic table:.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
